 Starting With “Hunt And Peck” Is Not The Way To GoĬonfronted with an IR spectrum of an unknown (and a sense of rising panic), what does a typical new student do? īottom line: The purpose of this post is to show you how to prioritize your time in an analysis of an IR spectrum. We’ll also see that there are at least two more regions of an IR spectrum worth glancing at, and thus conclude a “first-order” analysis of the IR spectrum of an unknown. See that forest of peaks from 500-1400 cm -1 ? We’re basically going to ignore them all!Ĩ0% of the most useful information for our purposes can be obtained by looking at two specific areas of the spectrum: 3200-3400 cm -1800 cm -1. With this in mind, we can simplify the analysis of an IR spectrum by cutting out everything except the lowest-lying fruit. In this way it’s complimentary to other techniques (like NMR) which don’t yield this information as quickly. Instead, IR is great for identifying certain specific functional groups, like alcohols and carbonyls. We don’t need to analyze every single peak ! (as we’ll see later, that’s what NMR is for : – ) ).For example, there isn’t a person alive who could look at the IR spectrum above and deduce the structure of glucose from it. IR is not generally used to determine the whole structure of an unknown molecule.In fact, once you learn what to look for, it can often be done in a minute or less. In this post, I want to show that a typical analysis of an IR spectrum is much simpler than you might think. Let’s Correct Some Common Misconceptions About IR Glucose, Revisited: The 1 Minute Analysisġ.Less Crucial, But Still Useful: Two More Very Diagnostic Areas.Specific Examples of IR Spectra of Carbonyl Functional Groups.Alcohols and Carboxylic Acids: More Detail.The Two Main Things To Look For In An IR Spectrum: “Tongues” and “Swords”.Starting With “Hunt And Peck” Is Not The Way To Go.Let’s Correct Some Common Misconceptions About IR.Your first impression of looking at that IR might be: agh! how am I supposed to make sense of that? We saw that the IR spectrum of water was pretty simple – but moving on to a relatively complex molecule like glucose (below) we were suddenly confronted with a forest of peaks! The result is a technique known as Infrared Spectroscopy, which is a useful and quick tool for identifying the bonds present in a given molecule. We can observe and measure this “singing” of bonds by applying IR radiation to a sample and measuring the frequencies at which the radiation is absorbed. These vibrations have frequencies that are in the mid-infrared (IR) region of the electromagnetic spectrum. Last post, we briefly introduced the concept of bond vibrations, and we saw that we can think of covalent bonds as a bit like balls and springs: the springs vibrate, and each one “sings” at a characteristic frequency, which depends on the strength of the bond and on the masses of the atoms. 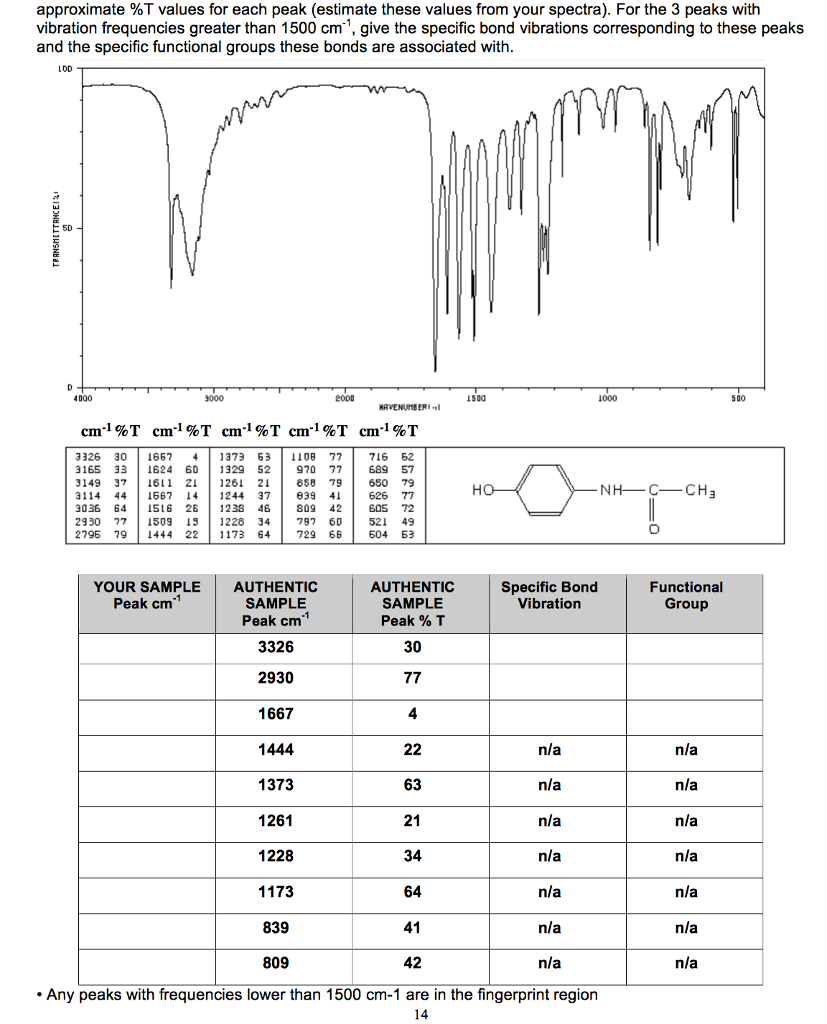
How To Interpret IR Spectra In 1 Minute Or Less: The 2 Most Important Things To Look For 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
